
There is positive evidence of human fetal risk based on adverse reaction data from investigational or marketing experience or studies in humans, but potential benefits may warrant use in pregnant women despite potential risks. This medication may not be approved by the FDA for the treatment of this condition.Īn Emergency Use Authorization (EUA) allows the FDA to authorize unapproved medical products or unapproved uses of approved medical products to be used in a declared public health emergency when there are no adequate, approved, and available alternatives.Įxpanded Access is a potential pathway for a patient with a serious or immediately life-threatening disease or condition to gain access to an investigational medical product (drug, biologic, or medical device) for treatment outside of clinical trials when no comparable or satisfactory alternative therapy options are available.Īdequate and well-controlled studies have failed to demonstrate a risk to the fetus in the first trimester of pregnancy (and there is no evidence of risk in later trimesters).Īnimal reproduction studies have failed to demonstrate a risk to the fetus and there are no adequate and well-controlled studies in pregnant women.Īnimal reproduction studies have shown an adverse effect on the fetus and there are no adequate and well-controlled studies in humans, but potential benefits may warrant use in pregnant women despite potential risks. The following list of medications are in some way related to or used in the treatment of this condition.įor ratings, users were asked how effective they found the medicine while considering positive/adverse effects and ease of use (1 = not effective, 10 = most effective).Īctivity is based on recent site visitor activity relative to other medications in the list. Examples include venlafaxine and fluoxetine. These are not specifically approved for ADHD but commonly prescribed. Examples include atomoxetine ( Strattera), and guanfacine ( Intuniv). They may be prescribed if stimulants are ineffective or have side effects but take longer to work (>3 weeks). Non-stimulants work by increasing norepinephrine levels.These are controlled substances with potential side effects and a high abuse risk. Examples include methylphenidate ( Ritalin, Concerta), amphetamine and dextroamphetamine ( Adderall) or lisdexafetamine ( Vyvanse). Available in immediate-release (short-acting) and extended-release forms. These are the most commonly prescribed treatments and work by increasing dopamine and norepinephrine levels. Pharmacological treatments for ADHD include stimulants, non-stimulants, clonidine, and antidepressants. 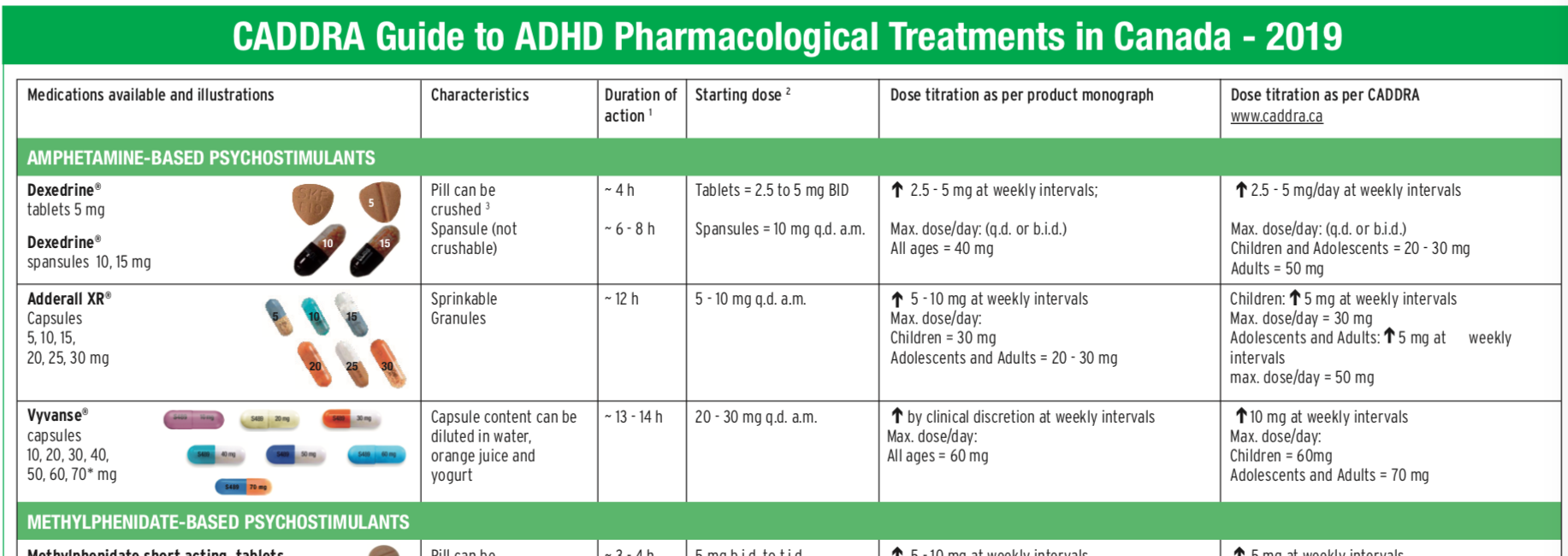
Behavioral education should be considered as the first-line treatment for any case of ADHD, particularly in children aged less than 6 years of age, with mild symptoms, or if the family prefers this option over drug therapy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
